THE CAUTI PROBLEM
Catheter-associated UTIs (CAUTIs) are still plaguing the medical community today. Current infection reducing solutions and protocols are failing to completely reduce a patient’s risk from contracting an infection during their stay. Complications due to a CAUTI can lead to increased length of stay, and even in extreme cases, death.1
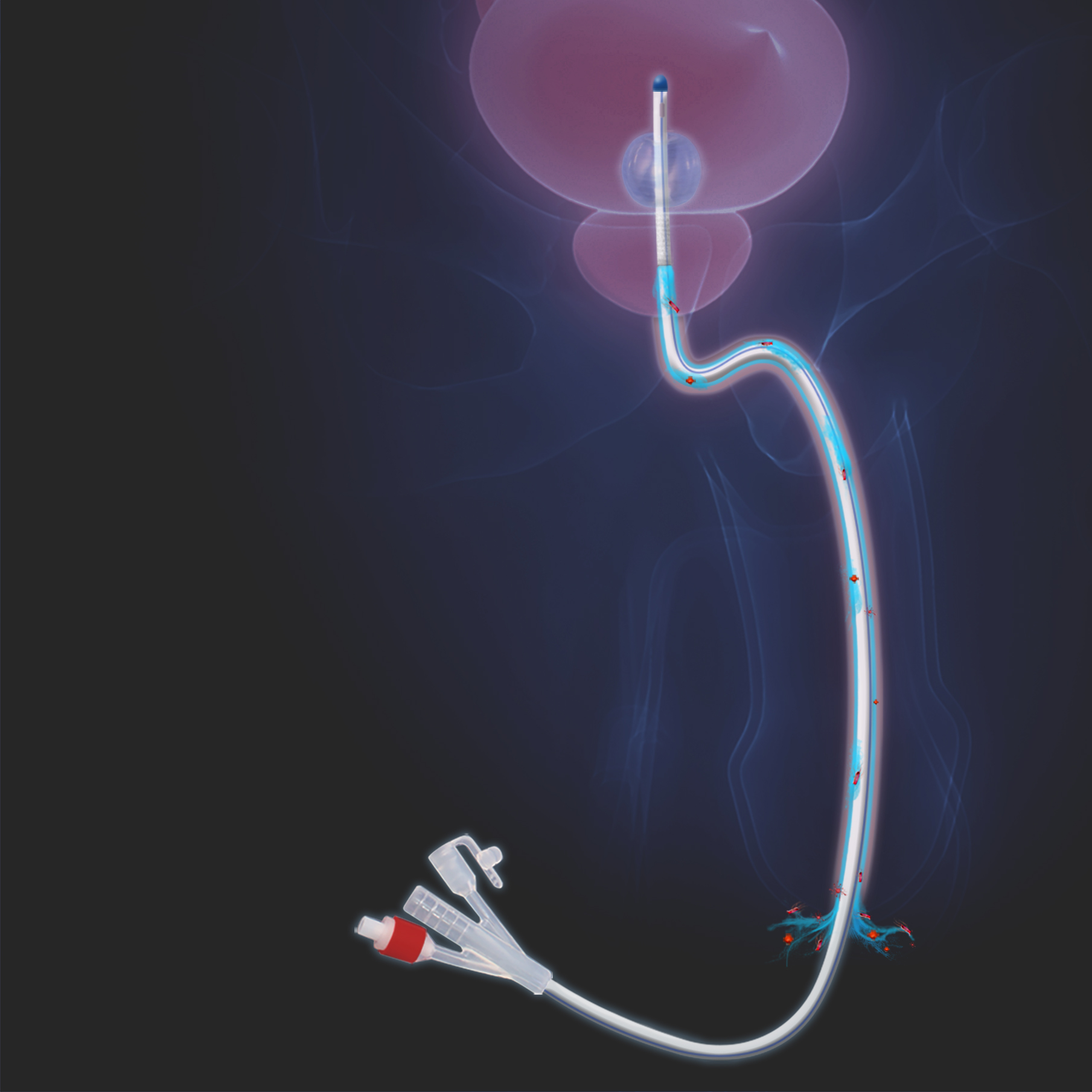
THE BLUCATH SOLUTION
The PSM BLUcath indwelling urinary catheter is developed to reduce bacterial growth and biofilm through an innovative transurethral flushing mechanism.
Using a flushing solution, BLUcath washes away harmful bacterial growth from the urinary tract as well as cleanses and hydrates the urethral membrane, all without the use of unnecessary antibiotics or other anti-infective agents.









 STUDIES
STUDIES